
Adverum Biotechnologies, Inc. (NASDAQ: ADVM)
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua.
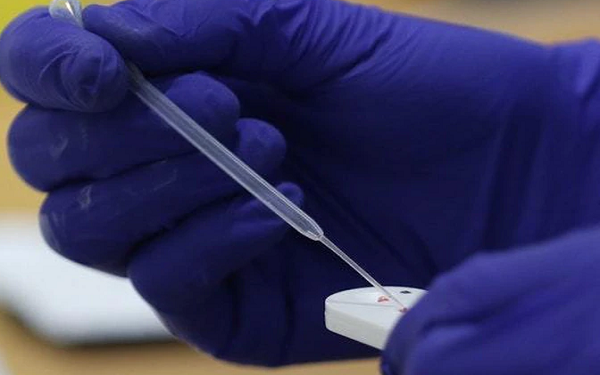
Abbott (NYSE:ABT) announces interim results from a multisite clinical study in urgent care clinics evaluating its rapid point-of-care SARS-CoV-2 molecular test performed on its portable ID Now analyzer.
The data showed a concordance rate with PCR-generated positives of at least 94.7% and a concordance rate with PCR-generated negatives of at least 98.6%.
ID Now identified all 29 positives and 226 of 227 negatives compared to Roche’s (OTCQX:RHHBY) cobas SARS-CoV-2 molecular test.
The system also demonstrated a 94.7% positive concordance rate and a 98.6% negative concordance rate with the CDC’s Novel Coronavirus (COVID-19) Real-Time RT-PCR Diagnostic Panel.
The company says the results reflect the expected performance of the system when used as designed, a direct reference to a recent small-scale study in New York that showed a much lower sensitivity rate.
Shares up a fraction after hours.
The British government has agreed to buy 10M COVID-19 antibody blood tests from Abbott (ABT -0.8%) and Roche (OTCQX:RHHBY -0.5%).
According to Health Minister Matt Hancock, the rollout will begin next week starting with healthcare workers and patients.
Pricing and contract terms are not disclosed.
Update: Abbott states that it has already shipped 800K IgG tests to the National Health Service (NHS). The assays can be performed on its automated ARCHITECT i1000SR and i2000SR and Alinity i laboratory instruments that can run up to 200 tests per hour. The NHS is already using its PCR tests to identify the presence of the SARS-CoV-2 virus from nasal swab specimens.
In a note, Evercore ISI’s Joshua Schimmer says Moderna’s (NASDAQ:MRNA) COVID-19 vaccine, mRNA-1273, is unlikely to be the best of the bunch but may wind up being “good enough” to get the first regulatory nod, adding that other candidates appear poised to offer higher immunogenicity, fewer (single) doses and less reactogenicity.
He also believes vaccine candidates from Vaxart (NASDAQ:VXRT) and Altimmune (NASDAQ:ALT) are “underappreciated” although investors’ lack of enthusiasm may be due to their timelines for clinical trials. The former will not start Phase 1 testing until H2 while the latter will not start until Q4.
Premarket action: MRNA up 1%, VXRT down 2%, ALT up 2%.
Other COVID-19 vaccine players: AstraZeneca (NYSE:AZN) (+3%), Sanofi (NASDAQ:SNY) (+1%).
CVS Health (NYSE:CVS) announces its plan to substantially increase the number of drive-through COVID-19 collection sites at selected retail pharmacy locations.
Georgia: 23 new sites.
New York: 36.
Maryland: 17.
Pennsylvania: 27.
Florida: 37.
Indiana: 21.
New Jersey: 11.
Texas: 44.
Massachusetts: 12.
Self-test swabs will be available to people meeting CDC criteria.
IMV (NASDAQ:IMV) is up 17% premarket after announcing positive preclinical results demonstrating robust immunogenic and antibody responses from the majority of peptide epitopes.
Based on these data, the Company has selected multiple peptide epitopes to be formulated within its DPX platform to form a vaccine candidate against coronavirus, DPX-COVID-19.
Majority of 23 peptide epitopes selected by IMV demonstrated robust antibody responses in an animal model after first and second dose in DPX formulation without any additional adjuvant.
DPX-COVID-19 is designed to focus the immune response on the weaknesses for the virus, enhance efficacy at preventing infection and reduce potential for immune escape.
A first-in-human Phase 1 clinical study of DPX-COVID-19 is scheduled to initiate during summer 2020.