With the rise of the Chinese middle class, “the love of beauty, the fear of death, and the lack of love” have become important psychological driving forces for consumers and while many sectors are seeing explosive growth, the health care industry now holds the most attention in the hearts of Chinese consumers. Companies with products related to healthcare, cosmetics and dentistry have experienced significant attention from both consumers and investors over the past months.
One such company is Milestone Scientific Inc. (NYSE AMERICAN: MLSS), a NYSE listed company which has developed and commercialized precise, efficient and virtually painless injection technologies for dental, epidural and arthritis treatment applications. Milestone Scientific’s first commercial application, called “the Wand®”, is now in dental clinics, where they help dentists safely, comfortably and precisely apply local anesthesia injections. The company’s dental products have been a commercial success and have entered 53 countries and regions, including Canada, Australia, Germany and China.
Aside from dental clinic products, Milestone Scientific also developed CompuFlo® Epidural, a computerized anesthesia system that has received 510(k) FDA clearance and a medical device license from Health Canada. The company’s next goal is to launch painless and precise injection equipment for botulinum toxin (botox) for the cosmetics market with hopes of matching the success of the Wand® in the dental industry.
The company has benefited from the expansion of its products into the US dental market and saw a 46.9% sales increase in the fourth quarter of 2017. The positive cash flow from the dental products has provided cash support for the marketing cost of other medical devices.
In the case of analyzing the development prospects of a company, we should first consider its existing and potential market size. Dental clinics have traditionally been a strong industry in the United States with its market size exceeding 130 billion dollars in 2017 and still rising steadily. However, in China, dentistry is still a fast-growing emerging industry. According to the Analysis of Chinese Dental Care Market Space in 2015, it is expected that the size of the Chinese dental medical device market was up to RMB134.6 billion yuan in 2015, and the overall size of the Chinese dental medical device as well as related products and services market will exceed RMB400 billion yuan. Meanwhile, the company’s technology has been in the market for many years, with sales breaking through first in the United States. In the field of medical device and pain management, American technology is still considered as more advanced than that of China, leading to potential advantages in adoption in China.
To help quickly expand into the Chinese markets, Milestone has established a joint venture in China focusing on local dental clinics.
The dental device (single tooth anesthesia – STA) is available all over China nationally in hospitals, clinics and private practices. As of now, in China, it’s mainly used for the dental anesthesia market. The company anticipates another 20,000 – 30,000 new STA being used in China in the next 2-3 years.
In June 2017, Milestone Scientific received 510(k) FDA approval for the – CompuFlo® Epidural Computer Controlled Anesthesia System (“CompuFlo® Epidural”). The CompuFlo® Epidural provides anesthesiologists with the ability to digitally determine and program pressure at the needle tip, in real-time. The Dynamic Pressure Sensing Technology™ (DPS) allows the CompuFlo® Epidural to provide objective visual and audible in-tissue pressure feedback that enables anesthesiologists to identify the epidural space.
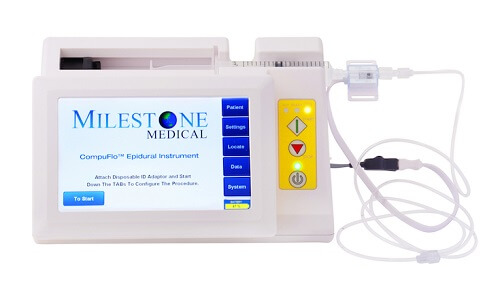
In March 2018, CompuFlo® Epidural received a medical device license from Health Canada. With both the US and Canadian markets open, the company has now moved onto the commercialization phase of their business. Leonard Osser, Interim Chief Executive Officer of Milestone Scientific, commented, “We are in discussions with distributors in North America and Europe. The trial results are expected to be published in major industry journals later this year, which will help to further accelerate product commercialization.”
According to the company’s marketing plan, the next goal after gaining a foothold in the dental and epidural treatment markets is to enter the medical cosmetic market. In 2018, the company, using its expertise in comfortable and programmable injection technologies, will introduce a medical cosmetic device for the injection of botox.
The development trend of the medical cosmetic industry is similar to that of the dentistry market, with its market consumption momentum expanding rapidly from the U.S. to China. In the United States, more than 8.4 million doses of botox are injected each year. According to data from Report on the Development Trend of Global Botulinum Toxin Market, the sales of global botox products reached USD$2.85 billion dollars in 2015, with the compound annual growth rate of 8.65% in the past five years. Similarly, China is a rapidly growing consumer market. Data shows that the market size of botox from formal channels in China in 2015 was between 1 billion and 1.5 billion with the compound annual growth rate of 27.32% in the past five years.
Milestone Scientific is now exploring opportunities to identify and engage strategic partners with the aim to commercialize the CompuFlo® Epidural and botox injection technologies in China’s dynamic markets.
Presumably, Milestone Scientific will first complete the commercialization of its products in the U.S. dental and medical cosmetic markets. The next step will be to enter into the emerging middle-class market in China to meet the “love of beauty, fear of death and lack of love” psychological demands of consumers.
More about Milestone Scientific’s investment highlights, recent press releases and presentation, please click here to visit Milestone Scientific’s company profile on NAI500.
Disclaimer: The company featured in this article is a client of NAI Interactive Ltd. This material is provided for information purposes only. It is not intended as a recommendation or an offer or solicitation for the purchase or sale of any security or financial instrument, or to enter into a transaction involving any financial instrument or trading strategy.